|
6/17/2023 0 Comments Hexane density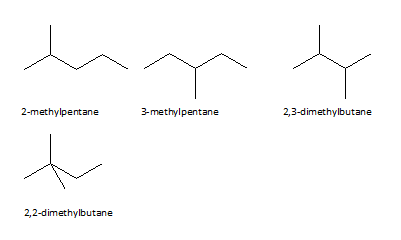
Hexanes are obtained from crude oil refining it is also prepared by various methods such as the reaction of propane with Na/dry ether forms hexane, hydrogenation of alkenes and alkynes also produce hexane. The structural isomers of hexane are given below. Hexane is categorized on the basis of its structure, their structure varies as the arrangement of carbon atoms. Ques: Does hexane have a double bond? Elaborate.Īns: Hexane is a saturated compound that contains single bonds of C-C and C-H bonds, unsaturated compounds have double or triple bonds.Īpart from this, Hexane molecular formula is C 6H 14 whose consists of 6 Carbon atoms and 14 Hydrogen atoms. Hexane has isomers that are formed on the basis of different arrangements of carbon as 2-methyl pentane, 3-methyl pentane, 2-2 dimethyl butane, etc.Īns: Hexane is used as a solvent, cleansing agent, and for extracting oils from seeds like peanuts, corn, etc.Īns: Yes, harmful effects are different in acute and chronic exposure, It may cause nausea, dizziness, headache and unconsciousness, etc. 
The other names of hexane are- Amyl Carbinol, 1-Hexanol, 1- Hydroxyhexane, and hexyl alcohol.It is a colorless, odorless, largely unreactive, volatile, non-polar solvent.Hexane is a straight-chain hydrocarbon having 6 Carbon and 14 hydrogen atoms bonded together with single bonds.Exposure to hexane by inhalation causes testicular damage in male rats.It affects the olfactory and respiratory epithelium of the nasal cavity of the mice. Long exposure to hexane in mice causes neurotoxic effects.Long-term exposure to hexane in the air causes muscular weakness, headache, blurred vision, fatigue, and polyneuropathy in humans.It may cause dermatitis and irritation of the eyes and throat in humans. Short-term exposure of high-level hexane to humans causes dizziness, nausea, headache, giddiness, and mild central nervous system effects.Hexane is also used as a cleansing agent in the printing industry and used as a liquid in low-temperature thermometers.It is used in the extraction of edible oils from seed and vegetable crops( soybeans, peanuts, corn) and used as a solvent for glues, varnishes, and inks.Comparisons to experimental results are discussed.Hexane comes under one of the most useful organic compounds which have certain uses in our daily lives such as. Our results signal the importance of molecular rolling and tilting (which result from the competition between molecule-molecule and molecule-substrate interactions) for the formation of the intermediate phase, while the insensitivity of the system’s melting temperature to changing density is understood in terms of in-plane space occupation through rolling. The solid-nematic transition temperature is very sensitive to coverage, whereas the melting temperature is quite insensitive to it, except for at low coverages where increased in-plane space and, ultimately, vacancies soften the solid phase and lower the melting temperature. As the temperature is increased, we observe a three-phase regime for ρ > 0.933 (with an orientationally ordered nematic mesophase) for lower coverages, the system melts directly to the disordered (and isotropic) liquid phase. If the density is decreased enough, vacancies appear. As the coverage is decreased, the amount of molecular rolling diminishes until ρ = 0.933, where it disappears (molecules become primarily parallel to the surface). The weight of unit volume of atmospheric air at given temperatures will be given later on. At high coverages, the solid consists of adsorbate molecules that are primarily rolled on their side perpendicular to the surface of the substrate. The density of hexane vapor is three times that of air. At low temperatures, the adsorbate molecules form a uniaxially incommensurate herringbone solid. We present the results of molecular-dynamics studies of hexane physisorbed onto graphite for eight coverages in the range 0.875 ⩽ ρ ⩽ 1.05 (in units of monolayers).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
